Multi-site QA & Negligible Risk Applications
Multi-site projects include any projects that involve more than one site, institution or centre. The definition of multi-site is where the Barwon Health HREC is asked to provide ethical approval for more than one site. Each site should have its own Principal Investigator and recruit/collect data from that site.
Multi-site Negligible Risk and Quality Assurance (QA) projects are reviewed by RDU without Committee Review so can be submitted at any time.
Read all of the points in the process (including information drop-downs) before commencing with your application. Note that the numbering system is arbitrary and that multiple steps can be undertaken in parallel to save you time.
If you’re not sure whether your research is suitable for a QA/ negligible risk application, please visit the how to prepare an application page or download and complete the ‘Level of Risk Checklist’. You may also contact [email protected] with any queries.
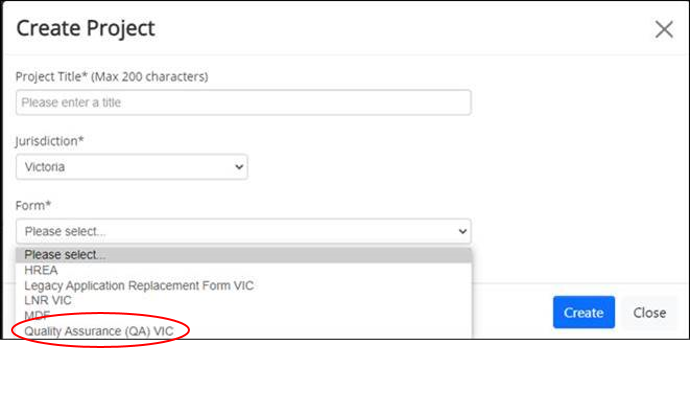
All application submission, feedback, correspondence, information requests and approvals occur within Ethical Review Manager (ERM). You must use the QA (Quality Assurance) VIC Form for submission of these research applications in ERM (do not use the HREA form):
- Negligible risk
- Clinical Audit
- Quality Assurance
- Evaluation Activities
Regional multi-site Negligible Risk/QA projects need to seek Barwon Health HREC approval/ exemption, then complete the governance steps below. Visit the Regional Health Service Collaborations page for a list of regional and rural sites that accept Barwon Health review.
Multi-site Negligible Risk/QA projects that intend to include metropolitan public healthcare institutions must first seek VIC metro reviewing site HREC approval, with Barwon Health listed as an approved site. Then follow the governance steps below.
Step 1. Generate a Barwon Health Reference Number
Generate a reference number for your project via the Barwon Health Research Reference Number Generator if you have not already done so.
Please ensure that your Barwon Health reference number is quoted at the time of your submission and included on the cover page of your protocol.
Step 2. Prepare ethics application documents
** Do I need to apply for a waiver of consent? Download and complete a waiver of consent checklist before you proceed, to avoid unnecessary paperwork.
Regional multi-site projects
- Quality Assurance (QA) VIC form – complete via Ethics Review Manager (ERM) – Select 'QA Form' from drop down 'Form' menu (include signatures from principal investigator, associate investigators, Head of Department)
- Protocol - click for template (download)
- List of all sites for which you are seeking approval
- Curriculum Vitae of all members of the research team - click for template
- Peer Review Form for student projects only - click for proforma and process (downloads)
Plus supplementary documents
- Waiver of Consent application - please complete the Waiver of Consent Checklist to confirm requirements (downloads)
- Authorship Record Form Template (download) - see Guidelines on Collaborative Research and Authorship
- Supporting Documents (surveys, questionnaires, etc.) - these can be set up in REDCap for secure electronic data capture
- Plain Language Statement (PLS) if using a survey/questionnaire
For step-by-step ERM guidance for Negligible Risk/QA submission see Page 74 of the Applicant User Guide to ERM
Multi-site projects including metro/national organisations
Multi-site Negligible Risk/QA projects that intend to include metropolitan public healthcare institutions must first seek external Victorian metro reviewing site HREC approval, with Barwon Health listed as an approved site. Then follow the governance steps below. Victorian metro reviewing sites include:
- Alfred Health
- Austin Health
- Melbourne Health
- Peter MacCallum Cancer Centre
- The Royal Children’s Hospital
- Monash Health
- St Vincent’s Hospital (Melbourne)
You need to contact the committee most suitable to your project for ethics review before proceeding with the Barwon Health application process.
Step 3. Prepare governance application documents (required for all regional & metro projects
All regional & metro projects
- Head of Department Signoff Declaration of support (download)
- If a Deakin University led study, an Annexure A is required (download). Please upload to ERM partially executed (with DU signature) and RDU will secure Barwon Health Executive signature once approved.
- Agreements with each participating site may be required. Visit the Barwon Health agreements page.
- Fee payment form – see Fees page to confirm requirements and applicability
Plus for metro projects
- All approved documents listed within metro HREC Approval/Exemption
Step 4. Submit your application
- Please refer to our document naming guidelines before submitting your application
- Ensure signatures are included for all investigators and Head of Department - see ERM signature guide.
- Submit your complete application (containing all supporting documents and signatures) via Ethics Review Manager (ERM) by clicking the ‘Submit’ button
Step 5. Review of your application
- Once submitted, your application will receive in office review. You will be prompted via email (address linked to your ERM account) to log into ERM and view any feedback, correspondence, and approvals relating to your application.
- If you receive a request for further information, you need to update the QA form and/or upload any missing documents and re-submit your application via ERM.
- Following a request for further information – if no re-submission OR correspondence is received from researchers within 3 months, the application will be withdrawn from ERM.
Contact RDU with any queries or concerns
RDU is here to help. If you are not sure where to start or have a question regarding your research project, email us at [email protected] and a member of the RDU team will get back to you within 24 hours. If you have a current application underway, please ensure you include your Barwon Health reference number in your enquiry.
Last Modified: Wednesday, 13 March 2024
